By Simone Ehrenberger, Institute for Vehicle Concepts, and Dr. Martin Tauber, IMA
Achieving zero CO2 emission levels has been a major driver for sustainability goals around the world, with many governments and companies declaring ambitious goals for a circular economy through 2030 and beyond.1 For large, traditional primary metal or processing industries, such as magnesium, being able to meet these deadlines requires immediate action, with investments and measures to be implemented immediately. For automotive and other OEMs to achieve their sustainability declarations (e.g., “Zero Global Impact” models), they need to take into account the sustainable sourcing of materials and components along the complete supply chain. Considering the global warming potential (GWP) of the complete vehicle and how its components are sourced is especially important as the automotive industry focuses on electromobility. Because of this, calculating the global environmental footprint through accurate life cycle assessment (LCA) is becoming an important decision tool for all stakeholders.
Transparent and Accurate LCA
When an automaker considers the sustainability and/or GWP of its vehicles, the OEM needs to consider the life cycle of all of the components within that vehicle, which are generally outlined in a bill of materials (BOM). A typical BOM consists of around 30,000 parts or components, representing a set of complete, transparent, and comparable information from Tier 1 and 2 suppliers. However, in order to achieve zero-emission sustainability goals, the OEMs also need to understand how raw materials (supplied to the Tier manufacturers) are sourced, which represents a greater challenge.2
A normal vehicle life cycle is divided into three main phases: production (including the sourcing and assembling), the use-phase, and end of life (when the vehicle is disassembled and landfilled or recycled). In all phases, international standards, databases, and statistics are available. However, there are still grey areas around the interpretation of the data and how guidelines are set for models, especially in regards to LCA analysis. For example, a common approach for the production phase is to divide the BOM list into material categories, allocating complete components to different materials. The data is then analyzed using GWP data from internationally recognized databases, such as GaBi or ecoinvent. It’s also common to separately analyze the raw material data from the batteries used in electric vehicles (EVs). This presents a challenge for lightweight materials like magnesium.
Magnesium is a lightweight champion among structural metals. However, in most of the recent vehicle LCAs, magnesium is allocated to an “other metals” category, which fails to recognize its potential. Although magnesium’s overall contribution to lowering GWP is limited due to it having a lower material content in vehicles (around 4.14 kg),3 the metal is expected to have a solid anticipated growth in future vehicles. As a result, it is important that magnesium be categorized separately, alongside aluminum, as another valuable light metal for achieving lightweighting.
Therefore, LCA databases should reference current and transparent data, with material flows and guidelines based on reliable data sources. When discussing other materials, such as aluminum, hundreds of different data points and values are generally presented. However, when discussing magnesium, many LCA studies and databases only offer a single primary average number for the Chinese Pidgeon process along with the electrolytic reference value. In addition, when comparing magnesium to the most common dataset for automotive parts made of steel, AAHS, aluminum, and CFRP, some studies rely on older references for magnesium that are based on outdated interpretations. Furthermore, the automotive move towards electromobility adds complexity to understanding LCA, in regards to both day-to-day sourcing and long-term material decisions.
All stakeholders need transparent and clear industry data. Such data sets are also necessary for future sourcing scenarios, enabling them to take into account aspects such as the different CO2 footprints of raw material in the production phase in relation to the material’s lightweighting potential in regards to the question of fuel or battery economy and the performance of vehicles in the use phase.
The International Magnesium Association (IMA) set out to address this challenge by commissioning the German Aerospace Center Institute of Vehicle Concepts (DLR) to put together an update of its 2013 LCA report. The new, updated study, “Carbon Footprint of Magnesium Production and its Use in Transport Applications,”4 provides a basis for transparent decision making and ultimately an answer to new consumer preferences. With focus on greenhouse gas (GHG) emissions, the study analyses the entire life cycle of magnesium components, including primary production, alloying, component production, use phase, and the end of life. Since magnesium production and especially the Pidgeon process in China is subject to continuous improvements, the new LCA study now more accurately reflects the current production situation. Furthermore, information on the GHG balance (expressed as CO2eq) of existing and planned magnesium production processes outside of China are included in the analysis. The study follows the international LCA standards, including DIN EN ISO 14040 and 14044 (ISO 14040 2006, ISO 14044) and was accredited by an external reviewer.
Primary Magnesium Production
The worldwide primary magnesium market has been dominated by Chinese producers for the last 20 years, representing an actual market share of around 85%. Until today, Chinese manufacturers have been mainly producing magnesium through thermal reduction, using the Pidgeon process. Other processes around the world include thermal-based processes in Brazil and Turkey, electrolysis production sites in Israel and China, and new methods being brought on stream in North America and Australia.
The Pidgeon Process in China
Several process steps are needed from raw material preparation to the actual reduction process to magnesium. Therefore, the cradle-to-gate assessment of magnesium production also includes its raw materials, in particular ferro silicon (FeSi), which accounts almost for 50% of all emissions in the process. The complete calculation for the GHG emissions from the Pidgeon process, includes dolomite mining, FeSi, fluorite supply, calcination, briquetting, reduction, and refining. Also, the different fuel gases used to power the refining process affect the environmental impact (Table I). According to the LCA system guidelines, environmental credits are calculated if the company reuses or recycles by-products that are produced during the process.
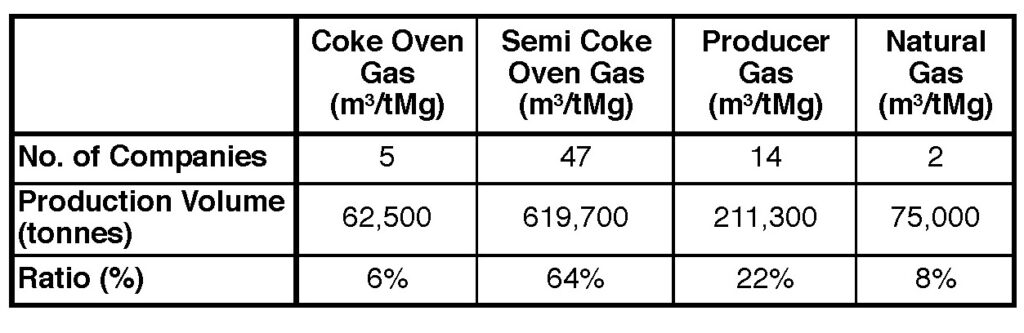
Over the years, the environmental impact of the Pidgeon process has been reduced, which is partly due to national and regional environmental policies being enforced by the Chinese government. Major efforts have been made to improve the energy efficiency of the process. In addition, stricter requirements concerning the reduction of air pollutants within China have forced magnesium producers to install additional equipment for air purification.
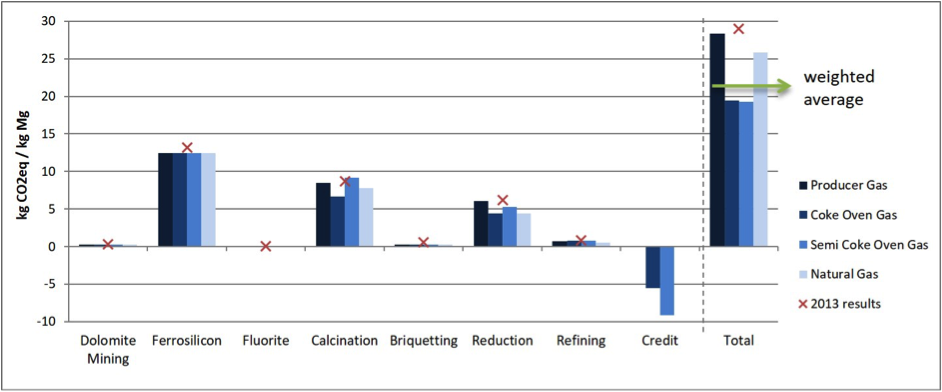
The weighted average emissions of the Pidgeon process from a cradle-to-gate perspective currently amount to 21.8 kg CO2 per kg magnesium. A detailed look at the GHG emissions for the various steps of the process, depending on different fuel gases, is shown in Figure 1. When alternative LCA system guidelines are used — excluding the FeSi emissions — the total emissions of the Pidgeon process are around 12.1 kg CO2eq per kg.
Other Processes on Industrial Level
Apart from China, the thermal reduction process is also used in Turkey and Brazil (Figure 2). In Turkey, Kar Madencicik operates a magnesium metal production facility, opened in 2016, using the Pidgeon process. Its CO2eq emissions are similar to those of the Chinese Pidgeon process. However, this has been considerably reduced through the operation of its own solar power unit, as well as potentially sourcing FeSi from suppliers that use a higher share of renewable energy.

In Brazil, Rima Group currently operates the only primary magnesium facility in the Southern Hemisphere. The production plant uses a silicothermic process, which is essentially a modified type of Bolzano process. Its calculated CO2eq emissions are 10.1 kg/kg Mg.5 This includes a credit for the CO2 uptake, as Rima Group has its own forests of eucalyptus trees, which are used for the production of carbon biomass.
In addition to thermal production, primary magnesium is being produced via electrolysis (Figure 2). In the case of electrolysis, the emissions depend mainly on the energy source used for this process. Dead Sea Magnesium (DSM) in Israel produces magnesium from carnallite, which is readily found in the Dead Sea brines. The analysis presented in the 2013 LCA study noted an overall GHG of 17.8 kg CO2eq/kg Mg, when no credits for by-products are given. The new study provides updated numbers and included credits for the processing of by-products for a GHG of 14.0 kg CO2eq/kg Mg.
Qinghai Salt Lake Magnesium Co., Ltd. (QSLM) also operates an electrolysis plant, which is located in Qinghai, China. In this process, pure magnesium is produced from magnesium chloride brine, which is a waste product of an adjacent potash production facility. The electricity supply of the magnesium production plant is mainly provided from hydropower. The overall GHG emissions from the electrolysis process amount to 8.5 kg CO2eq/kg Mg. However, the plant captures and reuses its chlorine emissions back into the production process, providing a credit that brings the overall emissions to 5.3 kg CO2eq/kg Mg. The results presented here are based on the assumption of full production volume. Since QSLM is still in its ramp up phase, it has not yet reached full capacity. Therefore, the current GHG balance would presumably show higher emissions per kg of magnesium, as a certain amount of energy is needed as a basic load for the plant independently from the actual production volume.
Planned Magnesium Production Projects
In recent years, various projects for establishing new production processes for primary magnesium have been planned. Due to the nature of such project-based processes being still in development, little information on the environmental performance is available. Nevertheless, generating low environmental impact in magnesium production is one criterion for establishing new primary magnesium facilities. This can either be achieved by using renewable energy, avoiding pollutant emissions, using waste from other industrial processes as raw material, or a combination of all three.
Figure 3 shows the GHG emissions of two different processes. Alliance Magnesium is developing a hydrometallurgical process in Canada combined with an electrolytic process using serpentine as raw material. Combined with a low carbon energy supply, the project is expected to achieve GHG emissions lower than 5 kg CO2eq/kg Mg.
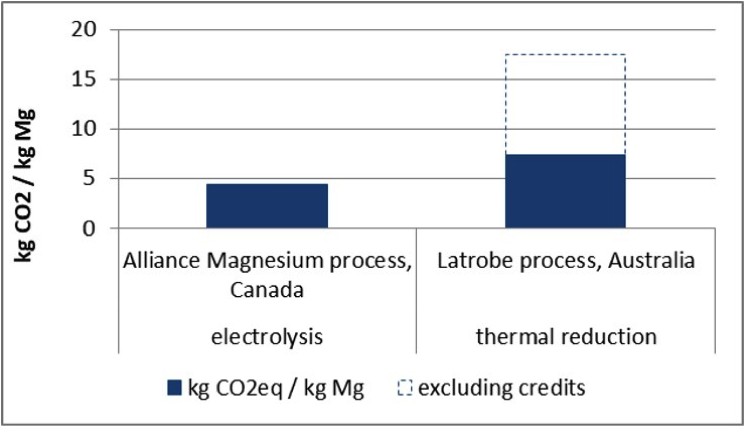
Latrobe Magnesium is another planned primary magnesium production site located in Australia, which will use fly ash, a waste material from brown coal power generation, as raw material. As a thermal process, the energy consumption is relatively high; the process as such has higher CO2eq emissions compared to the electrolytic process. However, the remaining ash waste generated from the process can be used as a cement substitute in the concrete industry. Therefore, credits given for the reuse of this by-product lower the emission balance to around 7.5 kg CO2eq/kg Mg.
Secondary Magnesium and End-of-Life Recycling
During the manufacturing and processing of magnesium components, scrap metal is generated. Some facilities will process this scrap in-house, but more often magnesium manufacturers will ship the scrap to dedicated magnesium recycling plants. Figure 4 shows the GHG emissions for two recycling plants run by Magontec GmbH in Europe, which were analyzed for the LCA study.
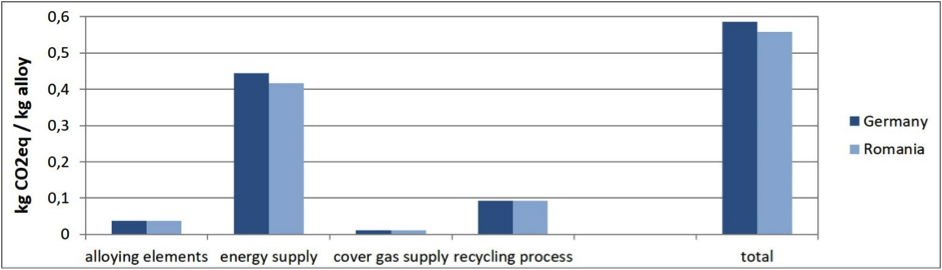
When looking at secondary magnesium and recycling, it’s important to consider not just the secondary production process itself, but also the origin of the scrap and how it is transported. This plays a major role in how the GHG balance is accounted for in secondary magnesium manufacturing. If the secondary material is made from production scrap, it basically adds up to being equivalent to the production process of the product. If end-of-life scrap is added to a product, it can replace primary material. Therefore, the use of secondary material and the calculation of its environmental burden need to be carried out transparently, so that they can be properly accounted for.
Technically, for end-of-life scrap, it would be possible to separate magnesium from the rest of the vehicle, but the relatively low volumes of magnesium per unit reduce the economic benefits of recovering the metal and less magnesium is recycled than possible. For the LCA, an automotive part was studied, taking into account substituting a functional and non-functional recycling of magnesium for primary magnesium in the follow-up process.
Case Study – Automotive Part
On average, magnesium shows higher emissions during component production compared to steel or aluminum on a per kg base. In general, these higher emissions tend to be compensated for during the use phase, as the amount of fuel and emissions that can be saved during vehicle use greatly depends on weight savings. Magnesium components can save about 25% of weight compared to aluminum. In this case, the sources of primary metal, as well as the end-of-life stage have a higher influence on the overall balance than in comparison to other products such as steel.
A similar logic might be applied to analysis of EVs. In this LCA study, a cross car beam made of AM50 magnesium alloy with a weight of 4 kg (Figure 5) was compared with the same part made of AlMg3 aluminum alloy with a weight of 5.4 kg. This comparison is based on the use of the component in a passenger car with a lifetime mileage of 200,000 km. The emissions from the die casting process (including alloying elements) were shown to amount to 1.5 kg CO2eq/kg Mg for the magnesium part and 1.4 kg CO2eq/kg Al for the aluminum part. It’s important to note that the scrap recovery rates for aluminum and magnesium are different (an update from the previous 2013 study). Aluminum has a scrap recovery rate of around 90%, while magnesium is at around 66%.3

A main contributor to emissions associated with the magnesium components is the use of primary metal. Therefore, the emissions range considerably between the different sources of magnesium (Figure 6). Due to the dominance of the Chinese Pidgeon process, the value is generally similar to the average Pidgeon process.

To fully understand the overall emissions for a magnesium component, however, the emissions from the overall life cycle need to be summed up. The results presented reflected a range of current probable scenarios, which are comparable to the European aluminum use mix. Just as with magnesium, if an aluminum part uses carbon-intensive material produced in China, the GHG emissions would be rather high. In scenarios in which a share of low-carbon secondary aluminum is used in the production of the part, the emissions drop considerably.
Equally, if upcoming magnesium production paths are compared to the aluminum component, the magnesium components could gain much higher emissions savings. Due to the variety of scenarios, general conclusions on the comparison of magnesium and aluminum parts cannot be drawn without ambiguity.
Conclusion
The IMA’s new LCA study, “Carbon Footprint of Magnesium Production and its Use in Transport Applications,” shows that, in general, there is a positive net balance of GHG emissions for all magnesium production scenarios that represent the current magnesium market. Since 2011, emissions from production of magnesium via the Pidgeon process have been reduced. Further reduction of the overall cradle-to-gate process emissions are also possible, e.g., when using FeSi from sources that rely heavily on renewable energy. An excellent example of how primary production can move forward is with the QSLM production site in Qinghai, China.
Another critical factor regarding the sustainability of magnesium is the use of secondary material. Both aluminum and magnesium have established pathways for recycling and reusing scrap from parts production, which is used for high quality secondary alloys.
The actual difference of emissions between magnesium and different products (such as aluminum, etc.) highly depends on the component characteristics and the material sourcing. Therefore, it is difficult to give generalized statements about the emission savings for these lightweight materials. To understand the full impact, the full life cycle of magnesium in automotive and aerospace needs to be considered, including the potential to reduce fuel consumption. In aircraft, especially the high fuel reduction potential leads to extremely fast amortization of emissions from the production stage. Therefore, the aircraft industry would likely see excellent benefits from the use of magnesium.
References
- “Z is for Zero Emission Vehicle (ZEV),” Volkswagen.
- “Being transparent about sustainability,” Polestar.
- “European Union Magnesium Recycling Study,” IMA, 2017.
- “Carbon Footprint of Magnesium Production and its Use in Transport Applications,” IMA, October 2020.
- Russ, D., J. Sandilands, and V. Hasenberg, “Dataset for Magnesium Production at Rima Industrial,” Leinfelden-Echterdingen: PE International, 2012.
Editor’s Note: This article first appeared in the February 2021 issue of Light Metal Age. To receive the current issue, please subscribe.
Simone Ehrenberger is a researcher at the department of Vehicle Systems and Technology Assessment at the Institute for Vehicle Concepts, which is part of the German Aerospace Center. Her research focuses on LCA of vehicles and vehicle components as well as on the assessment of tail-pipe emissions of passenger cars with alternative drivetrains. Before joining the Institute for Vehicle Concepts, she worked on various projects in the field of LCA at the Karlsruhe Institute of Technology as well as the research group for material flow and resource management at the Institute of Applied Sciences at Pforzheim University.
Dr. Ing. Martin Tauber has more than 25 years of experience in various basic industries, especially in the field of magnesium with management positions at large manufacturers, such as Norsk Hydro Magnesium and Magontec. In 2009, Martin Tauber founded FAURIS Management BV and has been offering worldwide services for the magnesium industry ever since. In the field of industrial representation, Tauber is the European Representative and former chairman of the European Committee of the International Magnesium Association (IMA) and president of the Critical Raw Material Alliance. He is also a recognized expert of the European Commission.