By Dr. Anne Deacon Juhl, AluConsult and AnodizingSchool.
Editor’s Note: Aluminum manufacturers are not always consistently satisfied with the surface quality of the anodized extrusion and sheet products returned from their anodizers. The anodized products may have patchy surfaces, color variations, or corrosion pits, causing rejects, higher cost, and frustration. Therefore, it is crucial that aluminum manufacturers understand the fundamentals of the anodizing process to communicate effectively with their anodizer. This series will provide an introduction to the overall anodizing process, including what properties are possible, which ones are right for a specific product, and how an aluminum company can find agreement with their anodizer on specifications in order to guarantee the correct results every time.
Anodizing is an electrochemical process, which thickens the naturally formed oxide layer on the aluminum surface. Growing the oxide layer from nanometers to microns in thickness, anodizing is able to change the properties of the aluminum product, resulting in higher corrosion resistance, coloring ability, and a consistent appearance. By changing the properties, anodizing makes aluminum more versatile for other applications, such as an anodized leg prosthesis that will not be scratched while climbing a mountain and will not be damaged by a swim in the sea.
There are several types of anodizing: chromic acid anodizing (Type I), sulfuric acid anodizing (Type II), and hard coat anodizing (Type III). For the purposes of this article series, we will be focusing on sulfuric acid anodizing (SAA), also called Type II anodizing, as it is the most common method and is used in 70% of all anodized products.
Benefits of Type II Anodizing
Anodizing provides a number of benefits for aluminum, increasing the product’s quality and versatility. This includes improved durability, excellent aesthetics, and sustainability.
Durability
The thickened oxide layer is almost as hard as a diamond, protecting the relatively soft aluminum material underneath. Since the anodic coating is essentially a conversion of the aluminum surface to aluminum oxide, it creates a chemical bond to the metal. Therefore, the surface layer is less likely to chip or peel. When processed correctly, most of the colors included in the porous layer will have a high level of UV and weathering resistance.
Aesthetics
The anodized layer is generally considered to provide an attractive appearance. On its own, the anodizing surface is transparent or “glasslike,” giving your product a high-quality surface, with no coloring needed. However, a beautiful range of colors can also be added to the anodized surface. This is possible because the anodized surface is porous, allowing dyes to be deposited into the pores, creating an attractive, long-lasting color.
Another benefit of anodizing is its metallic appearance, which highlights the aluminum itself. By comparison, a painted surface can create a more plastic-like appearance.
Sustainability
Anodizing is the most environmentally friendly surface finishing process — particularly when sulfuric acid anodizing and colors without chromium are used. By protecting the aluminum surface and preventing corrosion, anodizing extends the life of the aluminum product, thus decreasing its life cycle cost. Furthermore, anodized products can be directly recycled, unlike painted ones, which have to go through a pretreatment process to remove the paint and harmful chemicals.
The Anodizing Process
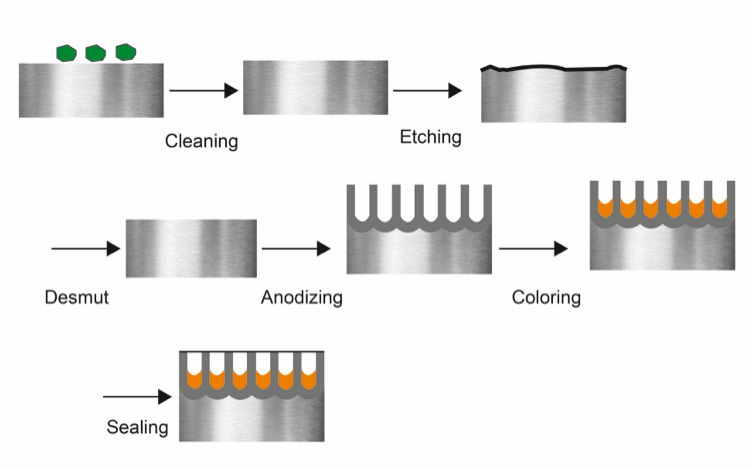
The anodizing process consists of several process steps and can be divided into three phases: pre-treatment, anodizing, and post-treatment. The most common method for Type II anodizing is shown in Figure 1. Note that the coloring step is optional.
Phase 1 – Pre-Treatment
To prepare the aluminum surface for anodizing, the metal needs to be cleaned. The most common process consists of three steps — degreasing, etching, and desmutting. Degreasing means removing any residuals from the surface that are caused by touching and machining the material. For better-than-best practice, the anodizer should perform a water-break test (a non-destructive method for measuring cleanliness).
Etching involves removing die lines and other surface defects from the extrusion or other semi-fabrication manufacturing processes. The most common etching process is alkaline. After this process step, the surface becomes smooth and has an even appearance.
During the etching process, some alloys will form black smut on their surface. Therefore, the last step of the pre-treatment process is desmutting, which removes the smut and prepares the aluminum surface for the acidic anodic solution.
Phase 2 – Anodizing
During Type II anodizing, the aluminum is submerged in a tank with electrolyte consisting of sulfuric acid and water. In the tank, an electrical current builds the anodic film on the aluminum surface, thereby forming a thicker artificial oxide layer with a porous structure. What happens in the anodizing tank changes the properties of the final product.
Therefore, to ensure the anodized parts match the product requirements, it is essential to discuss the desired properties with the anodizer and create specifications for the product. A later article in this series will discus alterations that can be done during the anodizing step to alter the properties of aluminum to match these specifications.
Phase 3 – Post-Treatment
At this stage in the anodizing process, the aluminum surface has a porous oxide structure. The porous structure is like a sponge that can absorb different materials in order to alter the surface properties. For example, the porous surface can be filled with glue particles to make it sticky. Likewise, colored dyes can be added to change the visual appearance. Finally, the aluminum is sealed, which closes the porous structure to create the highest protection of the final coating.
An important and undervalued process step is rinsing. This should be done repeatedly throughout the entire anodizing process in order to ensure a continuously clean surface and, thus, a flawless anodizing result for the aluminum product.
Alloy Microstructure and Its Effect on Anodizing
Outside of the anodizing process, the aluminum alloy itself has a great influence on the outcome of the anodized surface. Since the anodic layer is transparent, what you see on the surface before anodizing is what you will see after anodizing. It’s therefore essential to understand how the alloy can affect the results of the anodizing process.

To avoid varying surface appearances the aluminum should be measured using a conductivity devise or by microscope examinations. For example, Figure 2 shows micrographic images of an EN AW 6061 aluminum part. Though both images are taken from the same part, the microstructures in each is different — which will ultimately result in different appearance on the anodized aluminum part.
Keep in mind that the microstructure can vary not only from part to part, but even within the same aluminum product. This can be caused by machining or welding of the parts, which can cause different microstructures to occur in the same aluminum part. In other words, be aware of the metallurgical history of your aluminum parts and how this might affect the anodizing process and surface finishing.
Let me give a real-life example: in 2014, the design company Vitra wanted to launch the Landi chair (designed in 1938 by Hans Coray). The classic chair would go down in history as one of the first full-aluminum outdoor chairs. However, Vitra experienced difficulties with achieving the high-quality and aesthetic standard that their designs are known for. Specifically, the bent armrest caused the team great headaches (Figure 3), as it showed a grainy structure on the aluminum surface.

Vitra had almost given up on the Landi Chair when they reached out for a consultation. After three months of testing and analyzing the part, followed by further on-site analysis, Vitra and I were able to determine that the problem was in the microstructure of the aluminum arm of the chair. With this knowledge, the anodizer was able to alter the pre-treatment step to match the requirements of this particular aluminum alloy. Thus, the company was able to achieve the high quality surface required for its aluminum chair.
Conclusion
It’s vital that aluminum producers and end-users improve and maintain communication with their anodizers in order to achieve the surface finishing they desire. First, decide on the properties they want to achieve from the surface (appearance, resistance, versatility, etc.). Second, determine the overall properties that the aluminum component needs (strength, machinability, weldability, etc.). Tell your anodizer these desired properties and agree on process specifications and how to ensure the exact results. In other words, think about the surface properties first and the product properties later. Future articles in this series (available with a print or digital subscription) will go into more depth about how to achieve this by providing additional information on each of the process steps.
Editor’s Note: This article first appeared in the April 2021 issue of Light Metal Age. To receive the current issue, please subscribe.