Hospital-acquired infections (called nosocomial infections) are a common problem, causing the deaths of around 8-12,000 people per year in Canada and around 80,000 people per year in the U.S. A3 Surfaces, a start-up based in Saguenay, Quebec, Canada, is focused on making the world a safer place by eliminating cross contamination in healthcare institutions with high touch self-sanitizing fixtures and surfaces. The company has recently developed a groundbreaking surface treatment that can transform aluminum products into antimicrobial surfaces capable of significantly reducing the risk of bacterial and viral disease transmission (Figure 1), pushing cross-contamination from a matter of seconds to a few minutes (this means that it can break down tougher microbes that take more time to eliminate). In addition to use in hospitals, A3 Surfaces envisions its use in areas such as public transportation, large building HVAC for air intakes, classrooms, food conditioning, and more—potentially preventing the next pandemic.

Technology Development
Research on the technology began with an end-of-course project on aluminum transformation training at Chicoutimi College, when Maxime Dumont and Jocelyn Lambert had the idea of creating an antimicrobial aluminum. In the course of their research, they discovered that the application of an existing biocidal product on anodized aluminum had an antimicrobial effect—a concept that was submitted for a patent in 2013.
A3 Surfaces Inc. was founded by Martin Lambert along with co-inventors Maxime Dumont and Jocelyn Lambert in 2015 in Quebec, Canada, with the aim of further developing the potential of antimicrobial aluminum. During its launch, A3 Surfaces received financial and logistical support from Rio Tinto’s Regional Economic Development Team, which immediately saw the great potential of this technology. Over the past five years, Rio Tinto has invested a total of C$250,000 in start-up support to the company, facilitated connections with potential customers, and helped A3 Surfaces navigate regulatory processes with various levels of government.
Over time, A3 Surfaces has refined its biocide recipe in order to make it more effective and to produce a self-disinfecting anodized aluminum. The technology, named UmanProtek, allows aluminum parts that have received this surface treatment to be self-disinfecting. In December 2019, the company filed a second patent application for worldwide protection of the technology.
Currently, A3 Surfaces operates in a 1,100 sq ft leased space (Figure 2), where they have a chain of 13 production basins (3 ft long x 1ft wide x 2 ft deep). The current equipment is used for research and development work, as well as for the completion of current contracts for anodized antimicrobial parts. Processing of larger aluminum parts are subcontracted to an outside company that has anodizing baths of 8 ft x 3 ft x 4 ft. A production unit with 15 ft long baths is in the process of being built. The A3 Surfaces business model will be partly based on licensing because the anodizing market is specifically required to be close to the end users.

Antimicrobial Aluminum
UmanProtek is an elegant technology, merging the aluminum anodization process with biochemistry. Essentially, the biocide chemical release process that makes the technology possible is controlled by two laws of physical chemistry—Van der Waals force and Fick’s law of diffusion. Van der Waals is a weak force that actively takes place in the nano-world and enables the biocidal agent to be retained in the pores. Fick’s law controls the diffusion of the biocidal agent at the surface of the aluminum oxide substrate.
Choosing the correct, non-toxic biocide agent that would self-sanitize the surface was an important part of developing the UmanProtek technology. The biocidal agent had to meet several considerations, such as molecule size, diffusion rate, and chemical stability. In addition, if the biocide agent significantly affects the biota (friendly natural set of bacteria on people’s skin), it would create a harmful resistance to microbes. The agent also needed to be non-toxic and not create a resistance to antibiotics.
“We tested numerous biocidal agents and found that quaternary ammonium compounds (QACs) and silver were the best combination to inactivate Gram-positive bacteria, Gram-negative bacteria, and viruses,” said Jean-Luc Bernier, vice president of technology at A3 Surfaces. “The silver acts as a prophylactic agent, since it is not toxic enough to kill microbes, but is strong enough to avoid replications of microbes. Both QACs and silver have been used in health institutions for decades and approved by health authorities.”
The UmanProtek process is performed in four stages: surface preparation, anodization, impregnation of antimicrobial substances, and sealing. The surface preparation stage consists of cleaning and stripping the base material to get a uniform surface with minimum imperfections.
Anodizing type II and III are the critically important second step in the process, as they form the honeycomb of alumina nano reservoirs and control their aperture size. Hard anodizing (Rockwell hardness around 50) has to be monitored according to military (MIL) specifications. Most of the problems occur due to poor quality assurance. For instance, if an anodizer does not use deionized water in the washing steps, the water hardness will cause speckles, which create a visual appearance that is not acceptable.
Since some alloying elements, such as silicon, iron, etc., are detrimental to an uniform finish, not all aluminum grades are suitable to obtain the nanopores of the right size and aspect ratio. Therefore, 1000, 3000, 5000, and 6000 series alloys are preferred for uniformity of the nanopores and the potential of making end user products with a decorative and architectural finish.
The third stage is the impregnation of antimicrobial substances into the nanopores of the aluminum surface. The pores are filled with QACs, a type of antimicrobial substances commonly used in disinfectant wipes sold in grocery stores. During this step, the biocidal agent’s concentration and contamination have to be monitored.
Sealing is the final stage, in which the nanopores are closed. This proprietary sealing step has to be supervised carefully.
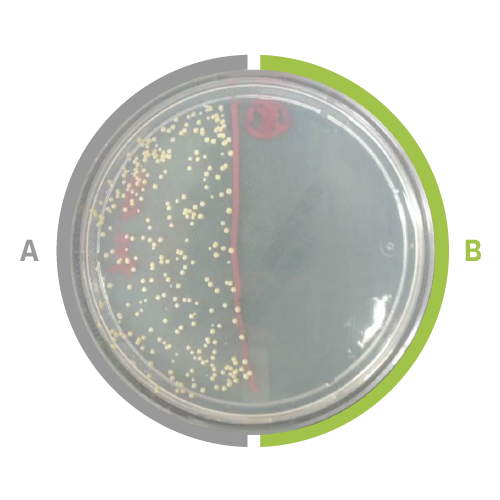
UmanProtek has been shown to significantly reduce the risk of transmission of microbial infections through cross-contamination, enabling aluminum parts to be self-disinfecting. Research performed by Dr. Nathalie Faucheux and Dr. Louis-Charles Fortier at the Université de Sherbrooke has found that products treated by A3 Surfaces can kill 99.9% of bacteria (S. aureus, a common cause of skin infections, respiratory infections and food poisoning) in five minutes or less (Figure 3). In addition, Prof. Nathalie Tufenkji from McGill University performed tests showing that the bacterial load (S. aureus) on a contaminated hand could be lowered by 80% by contacting the A3 Surfaces material for as little as five seconds.
It has also been shown that the antimicrobial aluminum treatment has a long lifespan, with the hard anodization and QAC agent providing long term stability. For example, a sample created ten years ago in the lab is still showing efficacy.
In addition, the National Research Council Canada (NRC) Aluminum Technology Center (ATC) completed abrasion tests to measure the wear with standard hospital cleaners. The ATC used a Gardner abrasion tester to perform 200 back and forths with a domestic sponge on the UmanProtek treated surface, with three different products: a bleach solution (0.5% active chlorine), Vert-To-Go disinfecting liquid (7 ml for 1 liter of water), and a stainless steel cleaner (undiluted). Results showed that the tested surfaces maintained their antimicrobial properties.
Moreover, A3S has a push plate installed in a public library, where 4,000 people per week are using the door plate. After 30 months, the bioefficacy is maintaining its initial performance. Based on these tests A3 Surfaces estimates that the antimicrobial aluminum treatment will have a life expectancy of 20 years.
Moving Forward
UmanProtek products from A3 Surfaces are now available in limited quantities. They can be sold within the U.S. under the exemption that covers products treated with EPA/FDA approved biocidal agents. For sales in Canada, the company is awaiting approval from Health Canada, which is progressing well.
Currently, large surfaces, such as walls or table tops have to be manufactured with aluminum extruded planks, which is uneconomical compared to continuous sheet coil anodizing. The company is in ongoing discussions with sheet coil anodizers in order to provide wider application of the process.
A3 Surfaces is working with the University Hospital of Chicoutimi and the Quebec Government to further demonstrate the technology’s efficacy in a hospital setting (Figure 4), with two rooms being monitored by an independent laboratory. One room is a standard hospital room with the typically used equipment. The second room was stripped down and renovated with A3 Surfaces biocidal aluminum parts including door handles, walls, trays, etc. The two rooms will be sampled weekly and the results will be compared, with surfaces tested to prove the reduction of the cross contamination. Although the experiment was temporarily stopped (partially due to COVID-19), the concerns have since been lifted and the experiment is continuing.

Rio Tinto will continue to provide financial and logistical support throughout the pilot testing phase of the anti-microbial aluminum technology. In 2020, the aluminum producer will focus on helping A3 Surfaces develop its business model and plans, as well as supporting its marketing efforts. “This Quebec innovation could be a major asset in the fight against epidemics and infections,” said Emmanuel Bergeron, director of Regional Economic Development Quebec. “It can have multiple applications in everyday life and we are very pleased to see the growing interest for antimicrobial aluminum.”
Editor’s Note: This article first appeared in the June 2020 issue of Light Metal Age. To receive the current issue, please subscribe.