By Jessica Elder, Worcester Polytechnic Institute,
and Jack Cowie and John Weritz, The Aluminum Association.
Although pure aluminum is utilized in many important products (such as electrical cables), alloying aluminum with other elements is crucial to achieve the mechanical, formability, and other performance specifications that are required for the use of aluminum in a majority of applications (including B&C, automotive, aerospace, packaging, and other markets).1 Each element is therefore crucial to the domestic aluminum supply base. The U.S. is highly dependent on foreign imports for many of these alloying elements. This article will demonstrate the need to reduce the country’s dependence on foreign sources of these aluminum alloying elements and the need to increase recycling as a way to retain them.
U.S. Production and Foreign Imports of Alloying Elements
The elements most commonly used for alloying include copper, manganese, magnesium, silicon, and zinc — however, approximately 30 elements are used to alloy aluminum.2,3 By reviewing information obtained by the U.S. Geological Survey and U.S. Materials Bureau, the Aluminum Association was able to determine the degree to which the U.S. is self-sufficient in producing these elements (Table I).4,5 In 2016, the U.S. produced 18 of the 30 identified elements commonly used in the aluminum alloying process, several of which were produced in small amounts. This analysis was based on the production versus import amounts, and from where these elements were being imported.
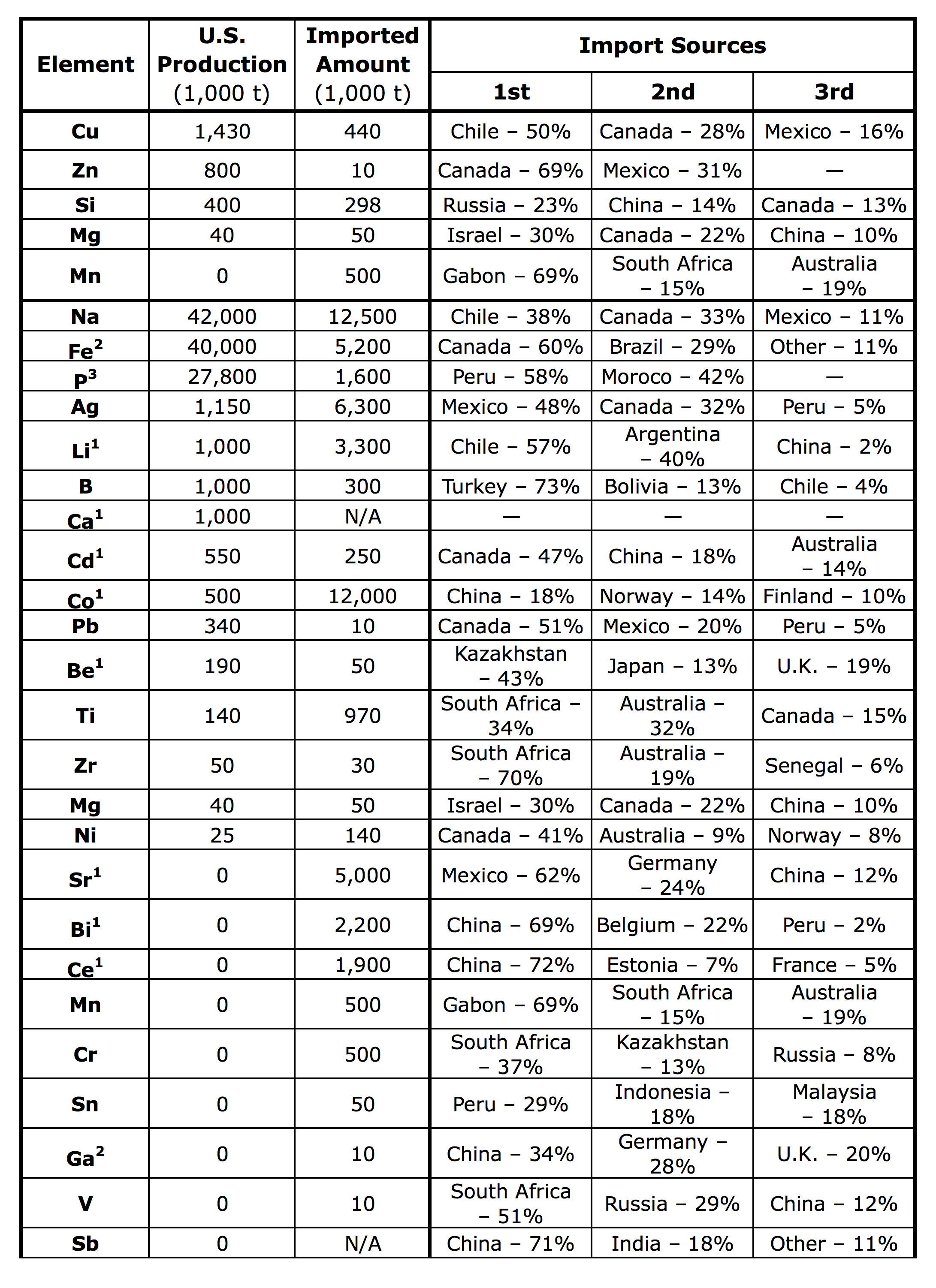
Some noteworthy data obtained regarding 2016 minerals in the U.S. and NAFTA region (the U.S., Canada, and Mexico) is as follows:
- The U.S. produced 1,000 tonnes of lithium out of 35,000 tonnes of the world’s production.
- The U.S. has no manganese reserves and no producing mines. North America is 100% dependent on manganese imports.
- The U.S. did not produce any bismuth, and the NAFTA region only produced approximately 550,000 out of 2.5 million tonnes.
- The U.S. did not produce any strontium, and NAFTA produced approximately 100,000 out of 350,000 tonnes of the world’s production.
- The NAFTA region did not produce any chromium (out of 34,000 tonnes), tin (out of 300,000 tonnes), vanadium (out of 75,000 tonnes), gallium, scandium, or cerium.
It is important to note that although the Geological Survey and Materials Bureau contained production information for each element, they lacked in-depth data specifically detailing how each element is used for alloy production. For example, although both sources contained production information for iron ore, the sources did not specify how much of this ore was being converted into a form used for aluminum alloys. While some elements can be alloyed with aluminum added in their pure elemental form, others have to be converted into specific forms—such as hardeners, briquettes, or grain refiners— that can more readily combine with molten aluminum.6 This poses a problem for the aluminum industry, as it is more difficult to find a suitable additive form of the exact chemical composition of an element required.
The U.S. Department of Homeland Security produced a list of strategic metals based on national defense, economic security, and public health and safety, as well as geological abundance. Other important factors that impact an element’s criticality include the potential for finding effective substitutes in production processes, the degree to which ore deposits are geopolitically concentrated, the state of mining technology, mining regulations, regional instabilities, and economic policies. This list of strategic metals included ferrous alloys, chromium, manganese, titanium, and cobalt, as well as minor metals germanium, niobium, tellurium, the rare earth elements, and tungsten. Notably, all of the top five metals on their list, plus cerium and scandium, are used in the production of commercial aluminum alloys.
Recycling
While limiting the use of elements in aluminum alloying cannot be easily changed, it is possible to reduce U.S. dependence on foreign imports of these elements through increased recycling of aluminum and other metal alloys. Alloying elements can be more easily reclaimed if the aluminum scrap is properly sorted by alloy series before recycling, so that the elements are not mixed.6 The best example of this type of recycling is aluminum used beverage cans (UBCs), which use alloys designed to streamline sorting and recycling so that the alloying elements do not vary from container to container. The can bodies are typically made from a 3104 aluminum alloy, while the lids are produced from a 5182 aluminum alloy.7 Both of these alloys contain many alloying elements that are listed as strategic metals, including silicon, iron, manganese, magnesium, and chromium, among others.
Aluminum UBCs offer a good example of how recycling can help reduce U.S. dependence on imported alloying elements. In 2016, the consumer recycling rate of aluminum UBCs was 49%, or 534,000 tonnes. At this rate, the amount of recovered manganese alone is 5,300 tonnes and the amount of recovered magnesium would be 4,600 tonnes. While a 49% recycling rate is considered very good for any commercial metal, it also means that just over half (51%) of all aluminum UBCs end up lost in landfills. This amounts to approximately 5,600 tonnes of manganese and 4,800 tonnes of magnesium lost per year. In the case of magnesium, this would account for more than 10% of U.S. magnesium imports each year.
Conclusion
Based on the analysis presented here, most of the elements alloyed with aluminum to provide their unique physical and mechanical properties are not mined in the U.S. The U.S. is highly dependent on Canada, China, South Africa, Russia, and South America to obtain all of the minerals needed for alloy production. In addition to this dependence, competition between different industrial sectors and different countries for the world’s resources is increasing. Without a reliable supply of these elements, many alloys could not be produced with their intended properties. These alloys are not only crucial for everyday life, but also U.S. national defense. Therefore, the domestic aluminum industry must have a secure source for all of these elements to ensure that it can properly produce all alloys required for a modern society. Given that aluminum alloys are used in defense related products for their unique properties, sudden supply interruptions of these alloying elements is an issue for the U.S. Department of Defense to consider in its strategic and critical materials studies. Increasing the aluminum alloy recycling rate as well as the recycling rate for other metal alloy systems could have a significant impact on U.S. dependence on imported strategic metals. This will become even more important as motor vehicle manufacturers increase their use of aluminum alloys.
References
- “History of Aluminum,” The Aluminum Association, www.aluminum.org/aluminum-advantage/history-aluminum.
- Aluminum Standards and Data 2017, The Aluminum Association, Arlington, VA, 2017, pp. 1-12.
- International Alloy Designations and Chemical Composition Limits for Wrought Aluminum and Wrought Aluminum Alloys, The Aluminum Association, Arlington, VA, 2015.
- “Mineral Commodity Summaries 2017,” U.S. Geological Survey, 2017, p. 202, https://doi.org/10.3133/70180197.
- World Metal Statistics Yearbook, 34th ed., World Bureau of Metal Statistics, 2017.
- Twarog, D., D. Apelian, and A. Luo, High Integrity Casting of Lightweight Components, North American Die Casting Association (NADCA), Arlington Heights, IL, 2016, pp. 54-67, 85-96.
- Hatch, John E., “Constitution of Alloys,” Aluminum: Properties and Physical Metallurgy, ASM International, 1984, pp. 47-53, DOI: 10.1361/appm1984p025.
Editor’s Note: This article first appeared in the December 2017 issue of Light Metal Age. To read more articles from this issue, please subscribe.